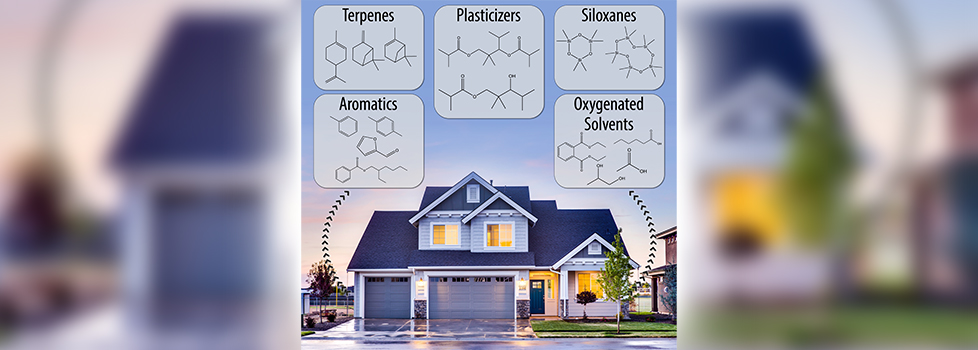
Check out our special symposium on “Coast to Coast Campaigns on Aerosols, Clouds, Chemistry, and Air Quality” at the 2024 AAAR Fall Meeting and consider submitting an abstract by May 8th. Hope to see you there!
Opportunities in the Gentner Research Group
I am looking for graduate students and postdocs with a strong science or engineering background who are passionate about the environment to join my research team. Please contact me if you are interested in working on big questions in air quality and global change.
There are also opportunities for select undergraduate students from Yale or with external REU funding.
Data processing and analysis code developed for complex mixture speciation, as well as the modeling of indoor air quality in recent publications can be found on github or archive pages associated with various publications (e.g., Ditto et al. ES&T Letters 2020, Gillingham et al. Science Advances 2021), or by contacting Prof. Gentner (e.g. for Agilent Q-TOF data processing code in Khare et al. J. Chrom. A 2019).